|
DEFEND FOR THE POOR CARBON |
|||
| 2.0 Antarctic Ice Core Data | |||
| 5.0 Heat Balance Mechanism | 6.0 Further Interpretation of Ice Core Data | 7.0 Coming Interglacial Peak Period | |
|
3.0 EFFECT OF GREENHOUSE GASES (GHG)
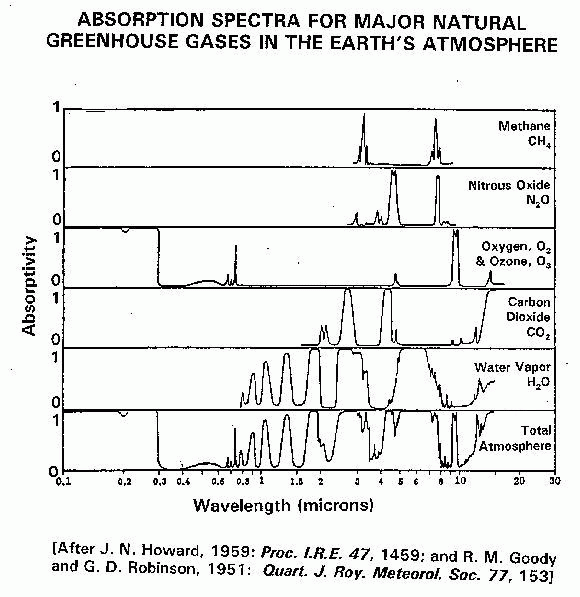
It was perceived that the global warming is caused by the increase of GHG, such as Carbon Dioxide, Ozone, water vapour, methane, nitrous oxide, Chlorofluorocarbon (CFC) and etcetera. Figure 3.1 and 3.2 below shows the absorption spectrum for varies GHG.[6],[7]
Figure 3.1 - gives the amount of energy absorbed by green house gases in various wavelength regions, from ultraviolet radiation on the left, to visible light in the middle, to infrared radiation on the right.
Figure 3.2 shows the absorption of ultraviolet, visible, and infrared radiation by various gases in the atmosphere for longer wavelength. Most of the ultraviolet light (below 0.3 microns) is absorbed by ozone (O3) and oxygen (O2). Carbon dioxide has three large absorption bands in the infrared region at about 2.7, 4.3, and 15 microns. Water has several absorption bands in the infrared, and even has some absorption well into the microwave region.
We take the plot for oxygen and ozone as an example; the absorption is very high in the ultraviolet region (below 0.3 microns or 300 nanometers) but essentially zero in the visible and infrared regions, except for isolated peaks. It is interpreted that this gas absorbs essentially all radiation in the ultraviolet but transparent in the visible and mostly transparent in infrared portions of the spectrum. Oxygen and ozone is responsible for shielding earth-based biological systems from ultraviolet radiation, but allows visible light and infrared radiation to pass through without much absorption.
Other gases have much different absorption properties. Methane (CH4) has a couple of very small wavelength regions in which it absorbs strongly and these occur at about 3.5 and 8 microns, which are in the infrared region. Nitrous oxide, N2O, having peaks at about 5 and 8 microns, absorbs in fairly narrow wavelength ranges. Carbon dioxide has a more complex absorption spectrum with isolated peaks at about 2.7, 4.3 and 15 microns.
The plot for water vapour shows an absorption spectrum more complex even than carbon dioxide, with numerous broad peaks in the infrared region between 0.8 to 10 microns, absorption well into the microwave region after 11 microns and fully shielded after 19 microns.
The total spectrum of all atmospheric gases is given in the bottom plot of figure 3.1 and top plot of figure 3.2. The total spectrum shows a “hole” between 0.3 and 0.8 microns (the visible region), which allows solar radiation (without UV component) to reach the earth's surface. The second “hole” at 8 and 10 microns are corresponding earth surface temperature, where heat from the earth is released to outer-space via radiation.
Figure 3.3 shows the solar radiation spectrum. The spectrum of the Sun's solar radiation is close to that of a black body with a temperature of about 5,800 K, which is approximately the sun’s surface temperature. About half that lies in the visible short-wave part of the electromagnetic spectrum and the other half mostly in the near-infrared part. Some also lies in the ultraviolet part of the spectrum.[17] When ultraviolet radiation is not absorbed by the atmosphere or other protective coating, it can cause damage to the skin known as sunburn or trigger an adaptive change in human skin pigmentation.
Figure 3.3 - The solar radiation spectrum for direct light at both the top of the Earth's atmosphere and at sea level. These curves are based on the American Society for Testing and Materials (ASTM) Terrestrial Reference Spectra, which are standards adopted by the photovoltaic industry to ensure consistent test conditions and are similar to the light that could be expected in North America. Regions for ultraviolet, visible and infrared light are indicated.
Figure 3.4 shows the summary of the solar absorption spectrum of GHG.[18] The radiation maximum for the Sun and Earth occurs at two distinct wavelengths of radiation. Both the Earth and the Sun emit electromagnetic radiation that closely follows a blackbody spectrum. The temperature of the Earth is much less that of the Sun. For the sun, these emissions peak in the visible region and correspond to a temperature of ≈5500 K, it is about 0.5 micrometers. Emissions from the Earth vary following variations in temperature across different locations and altitudes, but always peak in the infrared, about 288K, approximately 10 micrometers.
The position and number of absorption bands are determined by the chemical properties of the gases present. In the present atmosphere, water vapour is the most significant of these GHG, followed by carbon dioxide and various other minor GHG. As light passes through the atmosphere, some is absorbed by gases with specific absorption bands. Additional light is redistributed by Raleigh scattering, which is responsible for the atmosphere's blue colour. Collectively these processes capture and redistribute 25-30% of the energy in direct sunlight passing through the atmosphere. By contrast, the greenhouse gases capture 70-85% of the energy in upgoing thermal radiation emitted from the Earth surface.
Figure 3.4 – The effect for both solar radiation and upgoing Earth thermal radiation (top panel), the absorption bands in the Earth's atmosphere (middle panel) and. Individual absorption spectrum for major greenhouse gases plus Rayleigh scattering are shown in the lower panel.[18]
Figure 3.1 to 3.4 show that the effect of carbon dioxide in blocking the heat rejection from the earth is only on certain specific wavelength and minimal. Similar to other GHG except water vapour, the heat blocking by GHG is minimal. Water vapour plays major role in absorbing heat from solar incident before reaches the Earth surface, which heats up the atmosphere and insulating the earth from the outer space is the water vapour.
|
|||